Define and Describe an Isolated System and Give Examples
A force that is not balanced by other forces. An isolated system is a thermodynamic system that cannot exchange either energy or matter outside the boundaries of the system.
What Is Thermodynamic System Open Closed Isolated With Examples
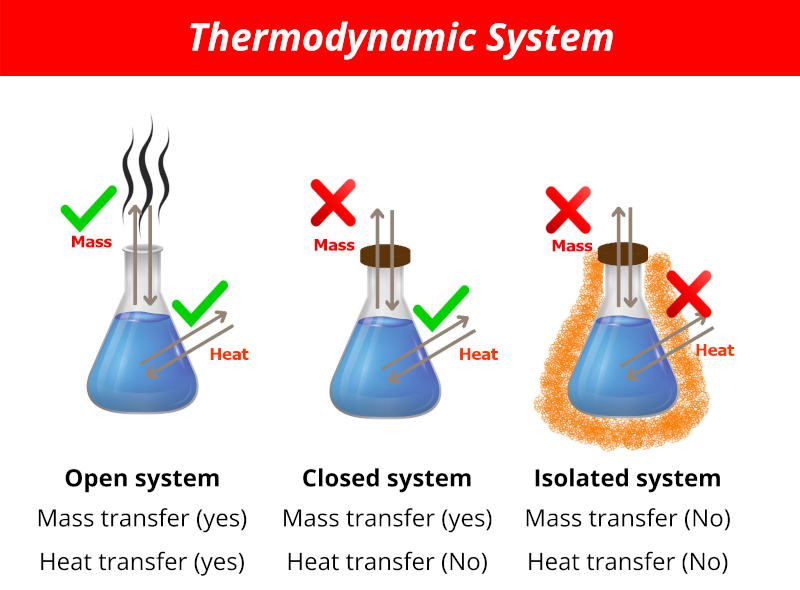
Isolated system is a system in which neither mass nor energy transfer takes place befween system and surroundings.

. An example of an isolated system can be seen by looking at the insulated thermos above. Cloistered covert hidden Antonyms. But I second the bomb calorimeter and the thermos.
A thermos flask But in reality this system does not exist because the hot water in this can not remains hot forever. A isolated system like a sealed thermos can exchange neither. Though subject internally to its own gravity an isolated system is usually taken to be outside the reach of external gravitational and other long-range forces.
Updated on December 07 2019. The main difference between open and closed system is that in open system matter can be exchanged with the surrounding whereas in a closed system matter cannot be exchanged with the surrounding. A closed system is one in which matter cannot go in or out but.
A closed system is one where a quantity or series of quantities cannot enter or leave the system. An open system is one which can allow mass as well as energy to flow through its boundaries example. Screened or sequestered from view.
A thermodynamic system enclosed by rigid immovable walls through which neither mass nor energy can pass. An isolated system is defined as in which the mass is fixed No mass can cross the boundary of the system and as well heat energy can not be transferred to its surrounding Example. A closed system like a closed water bottle can only exchange energy.
Isolated systems allow neither mass nor energy to flow through their boundaries. The system may be so distant from another system that it cannot interact with them. The concept of an isolated system can serve as a useful model approximating many real-world situations.
Isolated system and its example. This system is completely sealed. Practical example of isolated system will be thermos.
The only truly isolated system is the universe as a whole because for example there is always gravity between a system with mass and masses elsewhere. Neither matter nor heat can transfer to or from the surroundings. Basically if individuals cannot reproduce theyre considered to be different species and.
An open system like a cup of water can exchange matter and energy with the environment. A perfect isolated system does not exist as energy interaction in the form of heat radiation will always occur so. Law of Conservation of Energy Mass.
A system in which the only forces that contribute to the momentum change of an individual object are the forces acting between the objects themselves can be considered an isolated system. Hot tea remains hot inside it and cold ice remains cold inside it. Example A thermo flask.
All such thermodynamic systems where neither mass interaction nor energy interaction occurs between the concerned system and its surroundings are called isolated systems. For example a system might be. Universe is the only example which is perfectly isolated system.
A cup of coffee with a lid on it or a simple water bottle. Systems can be either opened closed or isolated. In these systems there is no way for matter or energy to be gained or lost.
Lastly an isolated system is a system that is completely closed off from outside interference. The car is a pretty straightforward example of an isolated system because its easy to see how you wouldnt move it by pushing from the inside or by pushing from opposite sides. Isolated System.
Total amount BEFORE process must EQUAL Total amount AFTER the. There are several types of reproductive isolation that lead to speciation. In an Isolated system neither energy nor masses are allowed to cross the boundary.
There are two ways in which this may occur. The purpose of a thermo flask is to keep your food warm. An isolated system is one in which neither matter nor energy can go in or out.
Systems can be either open or closed. Another good one might be a human body in an acclimated dry suit. Theoretically this is useful when constructing models.
An open cup of coffee. The system has fixed mass and energy. A closed system allows only energy transfer but no transfer of mass.
One of the most common methods is prezygotic isolation which takes place before fertilization occurs between gametes and prevents different species from sexually reproducing. A physical system so far removed from other systems that it does not interact with them. A closed thermos flask is an example.
I think its actually pretty hard to have a completely isolated system as there will always be some amount of heat or other energy transfer. Everywhere in the environment there are interactions between systems and their surroundings. Postby Thomas_Rodriguez_3G Thu Jan 26 2017 1022 pm.
No such system physically exists. Instead everything is kept constant. An example for an open system would be like an automobile engine or the human body.
However we can consider the whole universe as an isolated system considering there is no transferring of matter and energy outside the universe. Real systems may behave nearly as an isolated system for finite possibly very long times. Examples of isolated system.
For example the first and second thermodynamic laws describe an isolated system. A thermo flask can be considered an isolated system but only for a. Oceans with organisms gases and chemical compounds able to go in and out.
Frequent Find the right word. Example of isolated System.
Difference Between Adiabatic And Isolated System Definition Explanation With Examples
Open Closed And Isolated Systems With Examples Thermodynamics Psiberg
Open Closed And Isolated Systems With Examples Thermodynamics Psiberg
Belum ada Komentar untuk "Define and Describe an Isolated System and Give Examples"
Posting Komentar